Treatment
Use of Stem Cells in Orthopaedics
In the past several years, much has been written about stem cells, both in scientific articles and the general press. However, despite the amount of written material, there is still a lot of confusion about the exact role of stem cells in orthopaedic care.
This has, in part, to do with the rapid evolution of the understanding of the mechanisms of stem cells — basically, how they work — in specific orthopaedic conditions. The problem is made even more confusing by media outlets describing the use of stem cells in treating high-profile athletes.
To help you and your loved ones make decisions about whether stem cells may be an option for your specific orthopaedic condition — and whether they are likely to help you feel and/or function better — here are answers to frequently asked questions about stem cell therapy in orthopaedics.
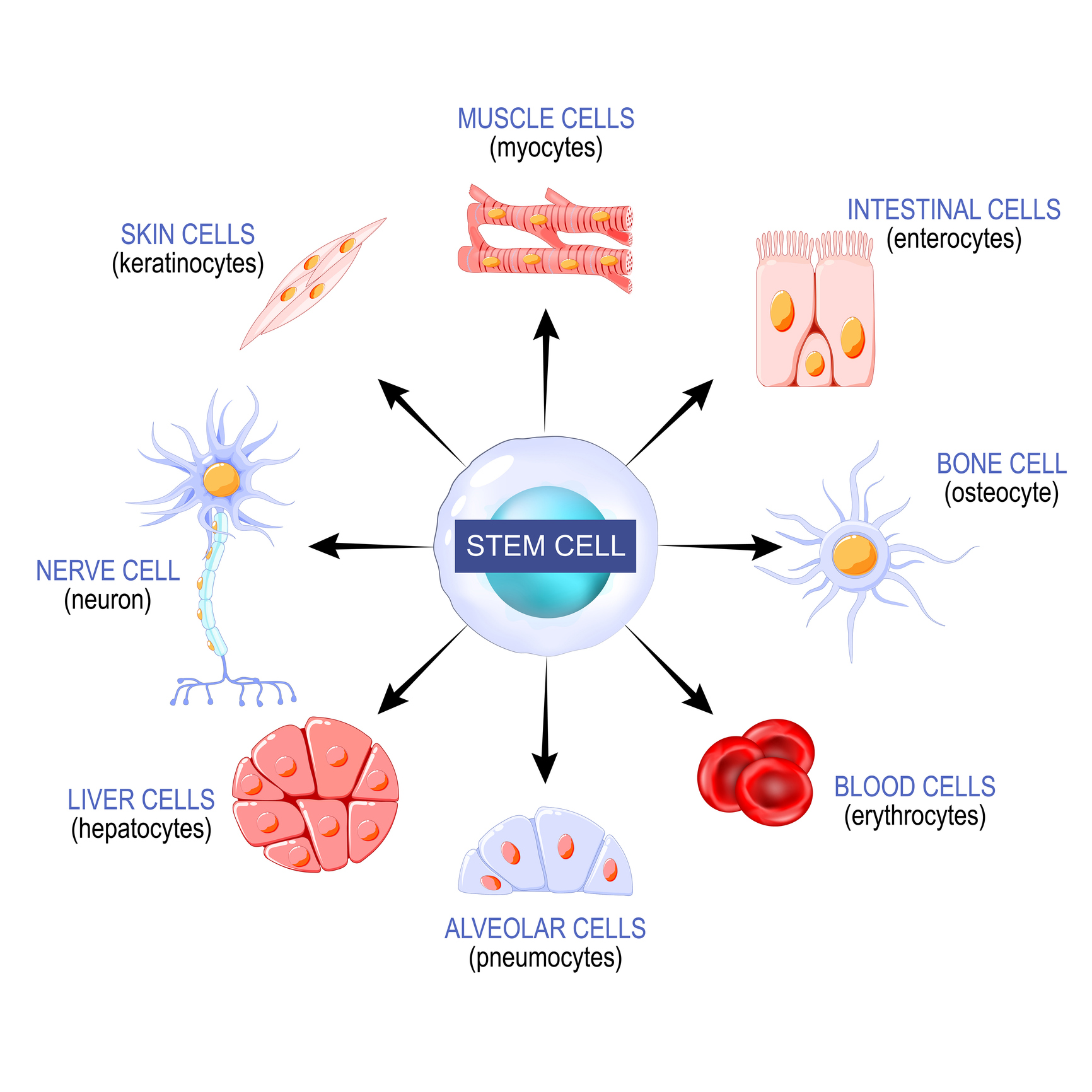
Q: What are Stem Cells?
A: Stem cells are the raw original cells of the body. These versatile cells form the building blocks of all cells: They are able to transform into all of the specialized cells your body needs, such as those that make up muscle (myocytes), bone (osteoblasts), cartilage (chondrocytes), and a variety of other tissues.
As many specialized cells mature, they lose their ability to replicate and heal tissue in people with certain diseases. However, because stems cells can retain this ability to replicate and heal even as they mature, it is thought that they could possibly regenerate damaged tissue.
An example would be a patient with damage to joint cartilage either because of trauma or degenerative changes over time (i.e., degenerative arthritis [osteoarthritis]).
- Current treatment for cartilage damage related to trauma is limited to harvesting and transferring cartilage from other parts of the patient's own body (autograft) or from donors (allograft).
- More advanced arthritis is usually treated with a joint replacement, in which the damaged areas of the joint are replaced with artificial (metal and/or plastic) parts.
Stem cells that could regrow the normal cartilage covering the joint would be a far superior treatment to those currently available.
Q: What types of stem cells are there?
A: There are several sources of stem cells:
- Embryonic stem cells. These stem cells come from human embryos that are typically 3 to 5 days old. These cells can divide into other embryonic stem cells, or into many other types of specialized cells. This versatility allows them to be used to regenerate or repair a wide variety of tissues. Access to embryonic stem cells is limited, however, and raises ethical and legal concerns. This restricts the use of these cells largely to research only.
- Adult stem cells. These cells exist in small numbers in adult tissues and can give rise to more limited types of body cells. While potentially not as effective as embryonic stem cells, adult stem cells can be harvested directly from the patient, so there is little risk of disease transmission and an immune reaction.
- Mesenchymal stem cells. These special adult stem cells can be grown in a lab and then converted to a category of tissues known as mesodermal tissues, which includes cartilage, muscle, fat, and bone. These types of tissues are obviously of special interest to orthopaedic surgeons.
Q: How do stem cells work?
A: While it was initially thought that stem cells could produce cells that would multiply and restore damaged tissues (like cartilage within the joint), there is little, if any, evidence that the injected stem cells survive long enough to multiply and create healthy new tissue.
It now appears that the potential benefit from implanting these cells comes from their ability to serve as "signaling cells."
What does that mean? Instead of doing the heavy lifting and building new tissue themselves, stem cells release "factors" that stimulate the body's existing stem cells (in the specific area where the stem cells are injected) to construct the new tissue. So, it is the patient's own stem cells that ultimately create the new bone, cartilage, or other tissues — at the urging and direction of the stem cells that have been implanted into the body.
Simply put, if you think of the injured body part as a construction site, the stem cells are the combined architect/foreperson, and the body's existing stem cells are the laborers.
Q: How does the FDA regulate stem cells?
A: While some stem cell clinics have stated that stems cells from one's own body are not regulated, the Food and Drug Administration (FDA) does, in fact, regulate the use of stem cell treatments to protect the safety of patients.
Stem cell treatments that come from one's own cells (autologous):
- Are only minimally manipulated while outside the body.
- Are used to treat tissues that are similar to the tissue from which the stem cell originates (homologous use)
- Are classified as human connective tissue products (HCTs).
These cells are felt to be safe for human use. However, these products must still meet basic standards for safety, such as sterility, potency, and freedom from contamination.
The FDA more strictly regulates and has required extensive testing prior to general use for stem cell treatments that:
- Do not come from the patient's own body
- Are concentrated, or are manipulated with drugs or other treatments
- Are used to treat substantially different types of tissues than the tissue of origin of the stem cells
Only a few of these types of stem cell treatments have been approved by the FDA. These can be accessed as part of clinical research trials that are testing and developing safe and effective stem cell therapies.
Q: What are the risks of stem cells?
A: Stem cell treatments categorized as HCTs are generally safe.
- As with any invasive procedure, infection can occur, even with appropriately prepared products.
- Worrisome reports of stem cell clinics that fail to adhere to FDA requirements for sterility and product preparation continue to be reported.
- Allergic and immune reactions are also possible.
Stem cell products not categorized as HCTs have significantly more risks.
- There are some reports of stem cell injections causing cancer at the injection site.
- With greater manipulation, the risk of contamination also increases.
Because of the increased risks, stem cell treatments that do not fall into the HCT category are strictly regulated. Multiple providers have been issued "warning letters" by the FDA stating that marketing of some providers' stem cell treatments not classified as HCTs is dangerous and illegal. The FDA is clear in their communications to patients that the patient who is a potential candidate for a stem cell treatment should get detailed information from their doctor about the FDA status of the treatment and associated risks.
Q: Is there strong evidence that stem cells are effective in treating orthopaedic conditions?
A: Despite decades of research, there is not yet strong evidence that stem cell treatments are effective.
- Given the strict standards of the FDA, only a few treatments that involve stem cells that do not qualify as HCTs have been approved.
- Stem cell treatments that qualify as HCTs are currently widely used. However, even HCTs only have mixed evidence of effectiveness in the available literature. Some articles have shown improvements in symptoms and function, but others have not.
Patients considering a stem cell treatment should carefully review with their doctor the available evidence about the effectiveness of the treatment and any risks involved. It is important to note that stem cell treatments are often not covered by insurance due to limited evidence supporting them, so patients may have to pay out of pocket.
Conclusion
- While the promise of stem cell treatments for orthopaedic conditions is significant, evidence supporting stem cell treatments is still lacking.
- Stems cells categorized as HCTs are widely available and generally safe; however, there is limited evidence of clinical benefit.
- Stem cell treatments not meeting the categorization of HCTs pose more risk, and very few products have been able to meet the rigorous criteria of safety and effectiveness required by the FDA. Potential risks of these types of stem cell are not insignificant, and access is possible only by enrolling in FDA-approved clinical trials, where the safety can be closely monitored.
Contributed and/or Updated by
Peer-Reviewed by
AAOS does not endorse any treatments, procedures, products, or physicians referenced herein. This information is provided as an educational service and is not intended to serve as medical advice. Anyone seeking specific orthopaedic advice or assistance should consult his or her orthopaedic surgeon, or locate one in your area through the AAOS Find an Orthopaedist program on this website.